What is GM1 Gangliosidosis?
GM1 Gangliosidosis (GM1) is a rare inherited genetic disorder that primarily affects children. It has many debilitating ramifications, some of which are developmental regression, mobility deterioration, seizures, visual impairment, and neurodegeneration. This is a fatal disease that currently has no cure or effective treatment. The thousands of diagnosed children and adults who suffer from GM1 face the progressive loss of skills and abilities until death.

This video provides a good overview:
The Cause

GM1 gangliosidosis is a lysosomal storage disorder (LSD). LSDs arise due to lysosomal defects. The type of LSD that exists depends on the lysosomal defect, but all of these defects cause errors in metabolism. These errors in turn result in the buildup of excess waste in the cells of the impacted organ or system. The accumulated waste then causes organ dysfunction, which can lead to chronic disease and death. Each lysosomal storage disorder is rare on its own, but collectively LSDs impact a significant number of people.
The genetic lysosomal defect for those with GM1 gangliosidosis results in a deficiency of the enzyme beta-galactosidase. This enzyme is needed to break down and recycle a number of biomolecules. The substance that accumulates most in GM1 gangliosidosis is called GM1 ganglioside. Abundant in neurons, GM1 ganglioside plays important roles in the nervous system, including in cell signaling, neuronal protection, neuronal recovery, and apoptosis. It’s also a point through which toxins, bacteria, viruses, and autoantibodies can enter cells. Without beta-galactosidase to break it down, GM1 ganglioside accumulates to high levels in cells and eventually causes cell death, predominantly neuron death. When enough neurons die, the nervous system progressively and significantly deteriorates, which eventually leads to death from complications of GM1 gangliosidosis.
References:
- NCBI Bookshelf: GM1 Gangliosidosis Overview
- FASEB Journal: GM1 Ganglioside and Lysosomal Storage
- Molecular Neurobiology: GM1 in Neurological Disease
Who Gets GM1 Gangliosidosis?
The inability to create a normally functioning form of the enzyme beta-galactosidase is due to a mutation in a gene called the GLB1 gene. This mutation is passed on via autosomal recessive inheritance. To understand autosomal recessive inheritance, it helps to understand the words that make up the term.
Autosomal
In genetics if a gene is autosomal, it means the gene is inherited in the same way by both males and females. In other words, the gene codes for a substance or characteristic that is not unique to male or female genders. The GLB1 gene is autosomal.
Recessive
Everyone has two copies of each gene, one from their mother and one from their father. Some genes are considered “dominant”, which means the trait they code for will be expressed, regardless of what the other gene in the pair codes for in terms of traits. Some genes are “recessive”, which means the trait they code for will only be expressed in the pair of genes, if the pair codes for the trait in a similar way.
Inheritance
This refers to something handed down from parents to children, and in genetics, that’s genes and traits. This means a person can only have GM1 gangliosidosis if both of their parents have a copy of the mutated gene and the copy of the mutated gene from each parent was passed down to their child.
Incidence of GM1
In the global population, only 1 in 250 people have a copy of the GLB1 gene with the disease-causing mutation. Their other GLB1 gene is normal. When a person only has one copy of the mutated GLB1 gene, they are called carriers. Carriers don’t have the disease but can pass down the mutated gene that causes it. Each carrier has a 50% chance of passing on the mutated gene. This means when both parents are carriers, their children have a 25% chance of inheriting two mutated GLB1 genes.
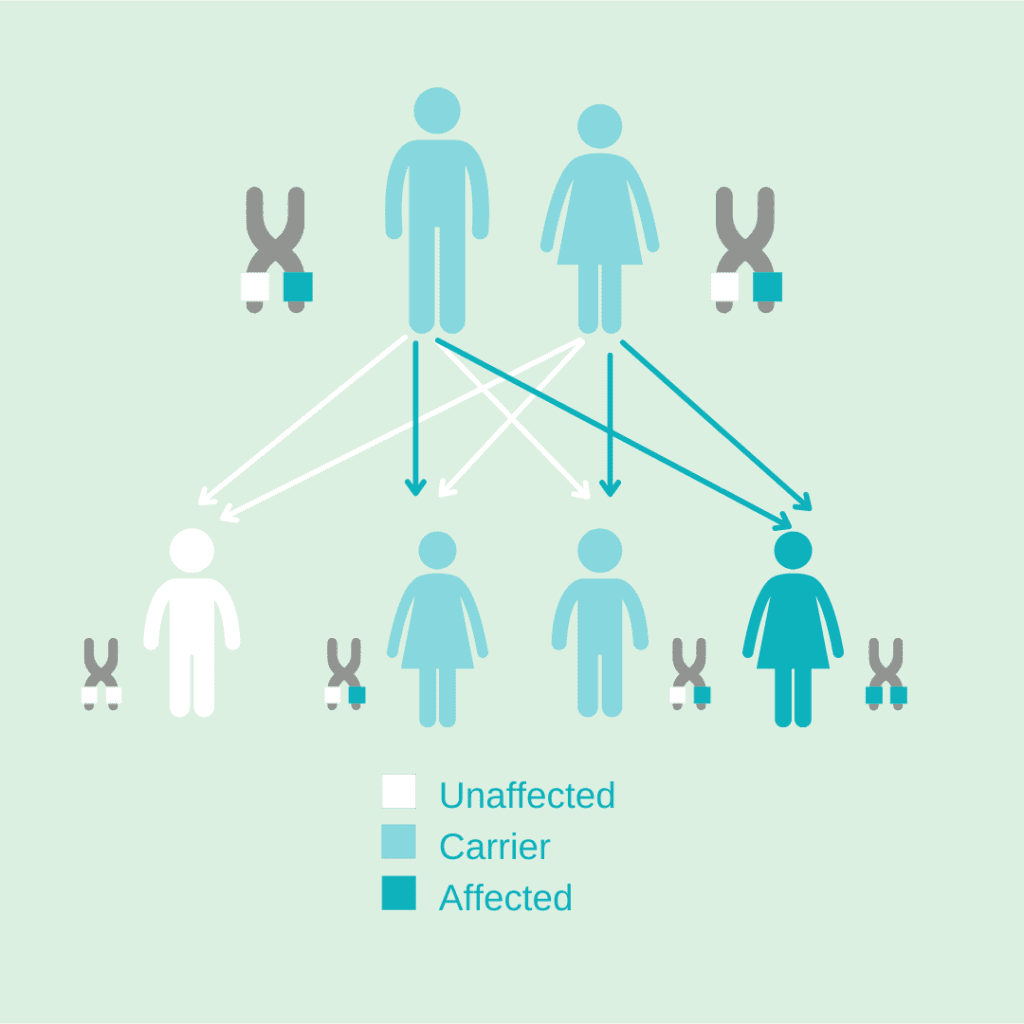
This is part of what makes GM1 gangliosidosis so rare. It is unlikely that two carriers have children, and if they do, they only have a 25% chance of having a child with the disease. Ultimately this means that only about 1 in 100,000 to 1 in 200,000 infants are born with GM1 gangliosidosis each year. However, the exact incidence varies depending on the source of the estimate. Newborn screening for GM1 is not yet universal, but advocacy efforts to add GM1 to newborn screening panels are ongoing. Learn more about newborn screening for GM1.
Subtypes & Symptoms
There are four subtypes of GM1 gangliosidosis: Infantile (Type 1), Late Infantile (Type 2a), Juvenile (Type 2b), and Adult onset (Type 3). Symptoms of GM1 gangliosidosis vary from person to person and type to type. Those with GM1 gangliosidosis may not experience the same symptoms as another patient with the same type of GM1, and the features of the different types can overlap to a great degree. Some symptoms that are typical across all types include: ataxia, dystonia, hypotonia, seizures, and loss of skills.
Symptoms of GM1 Gangliosidosis
Symptoms of GM1 gangliosidosis vary by subtype and by individual, but several features are common across all forms of the disease. Early signs may be subtle and are often mistaken for other conditions, which can delay diagnosis.
Common symptoms across all subtypes include:
- Hypotonia (low muscle tone)
- Ataxia (loss of coordination and balance)
- Dystonia (involuntary muscle contractions)
- Seizures
- Progressive loss of motor and cognitive skills
- Speech and language regression
- Visual impairment
Some children with the infantile form (Type 1) may also show coarsened facial features, enlarged organs (hepatosplenomegaly), and a cherry-red spot on the retina. Symptoms, severity, and rate of progression differ significantly across the four subtypes. Learn more about the subtypes.
History of GM1 Gangliosidosis
Tay-Sachs or GM2 Gangliosidosis was first identified in the late 1800’s. It was not until the 1930’s that that Tay-Sachs was characterized as an “inborn error of metabolism” and not until the 1960’s that the term “ganglioside” was introduced and GM1 Gangliosidosis was distinguished as its own disease.
GM1 Gangliosidosis is also known as Landing disease. Dr. Landing et al first identified the disease in 1964, calling it familial neurovisceral lipidosis.
Other Names and Identifiers
- Landing disease
- Norman-Landing disease
- Gangliosidosis-GM1 beta-galactosidase-1 deficiency
- Pseudo-Hurler disease
Life Expectancy for those Living with gM1
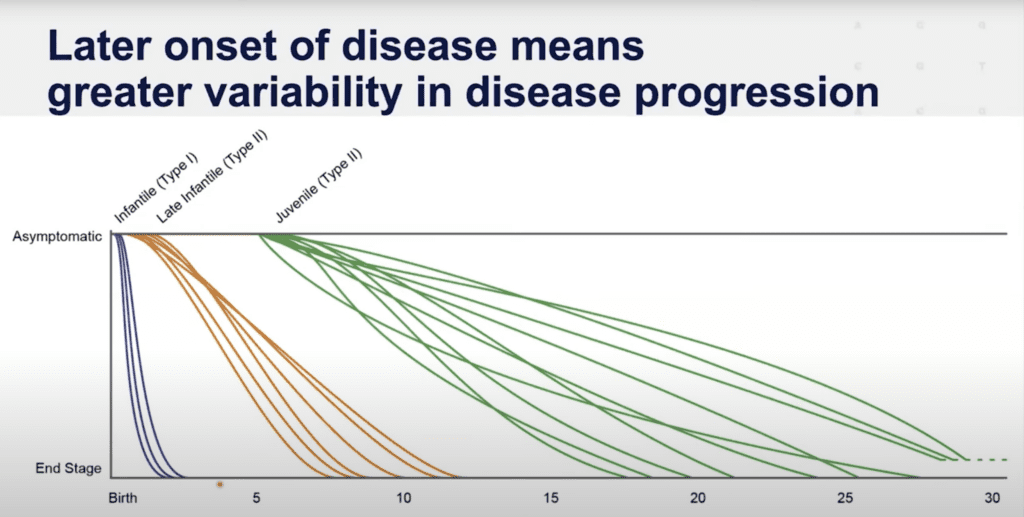
GM1 gangliosidosis is highly variable. Although there are distinct subtypes to organize various presentations of GM1, the disease is a spectrum based on the amount of residual enzyme. Many families ask why there are inconsistencies and a lack of predictability in life expectancy. No two exact cases are the exact same and many variables come into play such as the day to day medical care.
Different families make different choices with respect to the level of interventions, e.g., tracheostomy, feeding tube, participation in clinical trials, etc. Also, different publications represent different points of view. Researchers and authors see different groups of patients, therefore, the information and data with respect to life expectancy can also vary based on the population being studied. For example, the Middle East and Malta are known for having particularly severe infantile presentations of GM1, whereas the Japanese are documented to have higher incidence of of Type 3 / adult onset GM1. In general, life expectancy of adult-onset GM1 requires further study and is not in the diagram below due to lack of research and data.
Dr. Cynthia Tifft from NIH presented the following diagram in the GM1 Externally-Led Patient-Focused Drug Development meeting with FDA in 2022.
Treatment and Research
As of 2026, there is no approved treatment or cure for GM1 gangliosidosis. Symptom management remains the primary approach and may include anti-seizure medications, muscle relaxants for dystonia, physical therapy, occupational therapy, speech therapy, nutritional support, and palliative care. Research into disease-modifying therapies is actively progressing. The most advanced approaches include AAV gene therapy targeting the GLB1 gene defect, small molecule therapies such as pharmacological chaperones, and enzyme replacement therapy (ERT), which Cure GM1 Foundation is directly funding. An increasing number of investigational therapies have begun development in recent years. There is much to hope for, but a lot of work still to be done. You can help advance this research by donating.

We believe finding treatments and a cure is possible, we simply need to give researchers and drug developers the time and support they need to create them, which requires money. You can help advance this research by donating.
If you or a loved one are affected by GM1, you can find support in our Facebook groups. If you or your family are interested in participating in clinical trials and research, please contact us and we can answer questions. You can also see our natural history studies pages.
We invite and encourage anyone interested to take action to further our cause. There are many ways you can use your time and resources to make a difference. Your actions have impact.
To learn more, you can also visit our GM1 Gangliosidosis Facts page.
Frequently Asked Questions
What is GM1 Gangliosidosis?
GM1 Gangliosidosis is a rare inherited genetic disorder caused by a mutation in the GLB1 gene, which results in a deficiency of the enzyme beta-galactosidase. Without this enzyme, a substance called GM1 ganglioside accumulates in cells, primarily neurons, causing progressive neurodegeneration. It is fatal and currently has no approved cure or treatment.
What causes GM1 Gangliosidosis?
GM1 Gangliosidosis is caused by mutations in the GLB1 gene that lead to a deficiency of the enzyme beta-galactosidase. This is a lysosomal storage disorder, meaning the enzyme deficiency causes harmful substances to build up inside cells. The accumulation of GM1 ganglioside in neurons eventually leads to cell death and the progressive deterioration of the nervous system.
How is GM1 Gangliosidosis inherited?
GM1 Gangliosidosis is inherited in an autosomal recessive pattern. This means a child must inherit one mutated copy of the GLB1 gene from each parent to develop the disease. Parents who each carry one copy of the mutated gene are called carriers and typically show no symptoms themselves. When both parents are carriers, each pregnancy carries a 25% chance of an affected child.
How rare is GM1 Gangliosidosis?
GM1 Gangliosidosis is an ultra-rare disease, affecting approximately 1 in 100,000 to 1 in 200,000 infants born each year. Only about 1 in 250 people in the general population carry a single copy of the disease-causing GLB1 mutation.
What are the subtypes of GM1 Gangliosidosis?
There are four subtypes. Type 1 (Infantile) is the most severe, with onset in the first months of life. Type 2a (Late Infantile) has onset between 7 months and 3 years. Type 2b (Juvenile) has onset between 3 and 10 years. Type 3 (Adult onset) is the mildest form and may not become apparent until adulthood. All subtypes are progressive and currently have no approved treatment.
Is there a treatment or cure for GM1 Gangliosidosis?
As of 2026, there is no approved treatment or cure. Symptom management through medications, physical therapy, and palliative care remains the current standard of care. However, research is advancing. Cure GM1 Foundation is directly funding development of an enzyme replacement therapy (ERT), and additional approaches including gene therapy and small molecule treatments are under active investigation.
Ways to Give – Your Support Matters
RaiseRight | Walmart Spark Good | Facebook Fundraisers | Donate | Set up a Recurring Donation
Visit our Take Action page for more ways to support our community
